The Risks of Living with Long Term Gastroesophageal Reflux Disease (GERD)
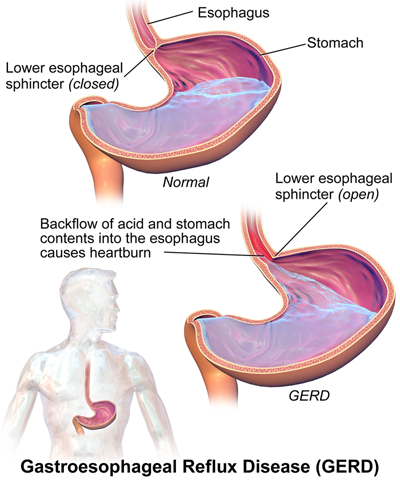 Reflux is caused by weak muscles in the esophagus called the LES (Lower Esophageal Sphincter.) The LES is the tool which acts as a barrier for stopping stomach acid from rising back up into the esophagus or throat.
Reflux is caused by weak muscles in the esophagus called the LES (Lower Esophageal Sphincter.) The LES is the tool which acts as a barrier for stopping stomach acid from rising back up into the esophagus or throat.
Although medications called Proton Pump Inhibitors (PPIs) can often control the symptoms of reflux by reducing the amount of acid in the stomach, these medications do nothing to actually address the cause of reflux.
Long term reflux disease, even symptomatically controlled by medications can have serious risks including Esophagitis, Barrett’s Esophagus, Esophageal Strictures and Esophageal Cancer.
Symptoms and chronic problems resulting from long term reflux may include heartburn, chronic sore throat, cough, shortness of breath, chest pain, asthma, difficulty swallowing, regurgitation, vomiting, bleeding, difficulty swallowing, dental erosion and bad breath, and hoarseness.
Esophagitis
When stomach acid rises up from the stomach repeatedly and contacts the lining of the esophagus, it causes erosions or a wearing away of the lining of the esophagus causing irritation, swelling, pain and even ulcers.
Barrett's Esophagus
As reflux disease continues over time, in about 10% of patients, a condition called Barrett’s Esophagus can develop. Precancerous changes to the lining of the esophagus occur which increase the risk of developing potentially fatal adenocarcinoma cancer of the esophagus. Around 1 in 300 people who develop Barrett’s Esophagus will develop esophageal cancer.
Dr. Merriman can diagnose Barrett’s by doing a test called an upper endoscopy or EGD. This is a scope which is placed down the throat to look at the lining of the esophagus and obtain a biopsy (tissue sample) which is sent to a pathology lab to be evaluated for abnormal cells.
Strictures
As the lining of the esophagus continues to experience damage from the reflux, scar tissue can form and build up causing strictures. These strictures can cause difficulty swallowing, eating and drinking.
The good news is a new revolutionary implant called LINX which is a titanium ring that acts as a natural enhancement to the LES described above. The LINX is placed laparoscopically around the outside of the esophagus. The LINX assists the LES in holding back stomach acid from flowing back up into the esophagus or throat. The device has sizes choses to fit your body and it is completely removable/reversible if necessary.
Dr. Merriman is one of only 2 surgeons in this area to be trained to do this surgery. He has placed multiple LINX devices and had excellent results in his patients. Click here to see the long term result patients have experience with the LINX device.
